Article Text
Abstract
Background Vitamin D deficiency during pregnancy is a public health problem in Pakistan and is prevalent among most women of reproductive age in the country. Vitamin D supplementation during pregnancy is suggested to prevent adverse pregnancy outcomes and vitamin D deficiency in both the mother and her newborn.
Methods We conducted a double-blinded, randomised controlled trial in Karachi, Pakistan to evaluate the effect of different doses of vitamin D supplementation during pregnancy on biochemical markers (serum 25(OH)D, calcium, phosphorus and alkaline phosphatase) in women and neonates, and on pregnancy and birth outcomes (gestational diabetes, pre-eclampsia, low birth weight, preterm births and stillbirths).
Results Pregnant women (N=350) in their first trimester were recruited and randomised to three treatment groups of vitamin D supplementation: 4000 IU/day (group A, n=120), 2000 IU/day (group B, n=115) or 400 IU/day (group C, n=115). Women and their newborn in group A had the lowest vitamin D deficiency at endline (endline: 75.9%; neonatal: 64.9%), followed by group B (endline: 84.9%; neonatal: 73.7%) and then the control group (endline: 90.2%; neonatal: 91.8%). Vitamin D deficiency was significantly lower in group A than in group C (p=0.006) among women at endline and lower in both groups A and B than in the control group (p=0.001) in neonates. Within groups, serum 25(OH)D was significantly higher between baseline and endline in group A and between maternal baseline and neonatal levels in groups A and B. Participant serum 25(OH)D levels at the end of the trial were positively correlated with those in intervention group A (4000 IU/day) (β=4.16, 95% CI 1.6 to 6.7, p=0.002), with food group consumption (β=0.95, 95% CI 0.01 to 1.89, p=0.047) and with baseline levels of serum 25(OH)D (β=0.43, 95% CI 0.29 to 0.58, p<0.0001).
Conclusion The evidence provided in our study indicates that vitamin D supplementation of 4000 IU/day was more effective in reducing vitamin D deficiency among pregnant women and in improving serum 25(OH)D levels in mothers and their neonates compared with 2000 IU/day and 400 IU/day.
Trial registration number NCT02215213.
- nutrient deficiencies
- nutritional treatment
- nutrition assessment
Data availability statement
Data are available in a public, open access repository. The datasets used for the article and the study is available from the corresponding author on request.
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
What this paper adds
Vitamin D level deficiency is a significant public health issue and is prevalent in more than 90% of women of reproductive age in Pakistan.
Due to adverse health consequences associated with vitamin D deficiency, particularly among infants, public health policy needs to be strengthened to ensure that vitamin D deficiency is minimised among pregnant women and their infants.
WHO and Institute of Medicine guidelines on vitamin D supplementation during pregnancy (200–600 IU/day) could be revised for populations of women at risk of deficiency in order to improve their vitamin D status.
More robust cohort trials need to be conducted to evaluate the effectiveness of optimal dose of vitamin D supplementation in populations with deficiency.
Introduction
Vitamin D has garnered a lot of attention in recent years due to a high global prevalence of vitamin D deficiency, which is affecting more than a billion people of all ages and ethnicity.1 The role of vitamin D has been recognised during pregnancy, such as for calcium absorption and regulation of placental calcium transport.2 Furthermore, maternal vitamin D deficiency has been commonly associated with adverse maternal and birth outcomes, such as vitamin D deficiency and hypocalcaemia in neonates, preterm birth, small for gestational age, and pre-eclampsia in meta-analyses and clinical trials.3–7
The prevalence of low vitamin D levels (<50 nmol/L or ≤20 ng/mL) in pregnant women is high in South Asian and Middle Eastern countries, particularly in India (96%), Bangladesh (64%), Turkey (90%), Kuwait (70%–83%) and Iran (60%–80%), where sociocultural and religious dresses prevent direct exposure of skin to sunlight, despite the abundance of sunshine throughout the year.8–12 The most recent National Nutrition Survey (NNS) 2018 in Pakistan reported a prevalence of vitamin D deficiency (≤20 ng/mL) of 79.7% in women of reproductive age, which has increased by 11%–13% since the previous NNS (2011).13 14 Another cross-sectional study conducted by our department found that 99.5% of women in Karachi and 97.3% of their neonates were vitamin D-deficient, possibly due to wearing of cultural dresses among women in the study which prevented them from direct exposure to sunlight.15 A recent study from a tertiary care hospital in Rawalpindi, Pakistan identified a prevalence of vitamin D deficiency of 61.5% among pregnant women.16
Limited trials have been conducted in Pakistan to evaluate the impact of different vitamin D supplementation on adverse maternal and neonatal health outcomes, where vitamin D deficiency is highly prevalent among the population. Moreover, the need for and the safety and effectiveness of vitamin D supplementation during pregnancy remain undetermined. There is no universal agreement regarding the appropriate dose of vitamin D during pregnancy, where it ranges from 200 IU/day to 600 IU/day (5–15 µg/day).17 18
However, studies have shown that vitamin D supplementation of up to 4000 IU/day is effective in obtaining sufficient serum levels of vitamin D (32 ng/mL) compared with 400 IU/day and 2000 IU/day in pregnant women and newborns and is safe for use.19–22 We conducted a randomised controlled trial among pregnant women to assess the efficacy and the pregnancy outcomes of different doses of vitamin D supplementation in our population, where vitamin D deficiency is very high among pregnant women.
Methods
Study design, setting and participants
We conducted a double-blinded, randomised controlled trial from June 2013 until December 2015 at Aga Khan University Hospital (AKUH)-affiliated Hospital for Women and Children, Kharadar in Karachi, Pakistan. Participant inclusion criteria were pregnant women less than 16 weeks gestation who had a singleton pregnancy and agreed to deliver at Aga Khan Hospital. Women with history of pre-existing chronic conditions (type I or type II diabetes, chronic hypertension or chronic disease), had multiple fetuses or had a fetal anomaly identified through an ultrasound scan were not eligible to participate in the study.
Sample size and randomisation
In order to detect a 40% reduction in vitamin D deficiency between the control and intervention groups, we calculated a sample size of 315 pregnant women in total (105 women in each group) at 80% power and 5% level of significance, based on a 68% prevalence of vitamin D deficiency among pregnant Pakistani women, according to the NNS conducted prior to our study.14 To account for dropouts and lost to follow-up, the total sample size was inflated to 350 women. The data management unit of Aga Khan University (AKU) created a block randomisation list with a block size of 10 to balance the number of participants allocated into the study groups.
Study intervention, blinding and allocation
Study participants were allocated into three groups of vitamin D3 supplementation: 4000 IU/day (group A), 2000 IU/day (group B) and 400 IU/day (group C). Group C served as the control. Vitamin D supplementation was started between the 12th and 16th week of gestation. The randomisation list was provided to the AKUH pharmacy, which prepared and packaged the supplements according to allocated dosage and unique participant identification. The supplements were distributed as oral syrups in bottles that were identical in shape, size, colour and taste. A 5 mL dose of the syrup contained 4000 IU, 2000 IU and 400 IU in groups A, B and C, respectively. The study staff (investigators, laboratory staff, study team and data collectors) and participants were blinded to the dose of vitamin D allocated to participants. The allocation scheme was made available to the pharmacy in cases where individual participants need to be unmasked due to suspected supplement-related adverse events (ie, hypercalcaemia or vitamin D toxicity).
Outcomes and measures
The primary outcome measures included vitamin D and calcium deficiency, pregnancy, and birth outcomes, that is, pre-eclampsia, gestational diabetes mellitus (GDM), preterm birth (<37 weeks gestation), low birth weight (≤2500 g) and stillbirths (no signs of life on delivery of baby). Based on clinical classifications, we defined severe vitamin D deficiency as serum 25 Hydroxyvitamin D (25(OH)D) concentration <8 ng/mL, deficiency as <20 ng/mL, insufficiency as 20–30 ng/mL and sufficiency as 30 ng/mL or greater.13 23 Pre-eclampsia was suspected through blood pressure (>140/90 mm Hg) and confirmed through a urine dipstick test or urine analysis for proteinuria (≥300 mg).24 GDM was diagnosed through glucose intolerance during an oral glucose tolerance test conducted in the second trimester of pregnancy. Glucose intolerance was defined by fasting glucose >92 mg/dL, 1-hour glucose >180 mg/dL or 2-hour glucose >153 mg/dL.25
Adequacy of sunlight exposure in the house was self-reported and was assessed based on whether the house had windows through which sunlight enters during the day. Moreover, we measured exposure to sunlight based on the time spent by the participant in sunlight.
Safety assessment
We monitored all subjects for adverse and serious adverse events, as well as for hypervitaminosis D (serum 25(OH)D >150 ng/mL) and hypercalcaemia (serum calcium >10.2 mg/dL).23 If a participant were to exceed the serum 25(OH)D limit or experienced any adverse event, the AKU Ethical Review Committee was immediately informed and the participant was treated accordingly by their clinician.
Enrolment and data collection
Pregnant women who provided written consent were enrolled in the study between the 12th and 16th gestational week. Baseline information collected at enrolment included socioeconomic measures and anthropometric measurements (weight, height and body mass index). We also documented gestational age, women’s exposure to sunlight, dietary patterns, and reproductive and medical history and examination. Baseline information was collected by trained data collectors. Gestational age was determined based on the last menstrual period and dating ultrasound. Diet was evaluated through consumption of 10 food groups 24 hours prior to visit. The food groups included the following: (1) grains, white roots, tubers and plantains; (2) pulses; (3) nuts and seeds; (4) dairy; (5) meat, poultry and fish; (6) eggs; (7) dark green leafy vegetables; (8) vitamin A-rich fruits and vegetables; (9) other vegetables; and (10) other fruits. Minimum dietary diversity was achieved if participants had consumed at least 5 of these 10 food groups in the 24 hours prior to their follow-up visit. Participants were provided with a supply of vitamin D which was replenished at follow-up visit. They were instructed to consume 5 mL (1 teaspoon) of vitamin D syrup a day, which was equivalent to their allocated dosage. Each bottle contained 180 mL of vitamin D syrup with markings at every 5 mL to indicate daily dosage requirements.
Follow-up visits were conducted monthly until 28 weeks of gestation, fortnightly until 36 weeks and then weekly until the time of delivery. During visits, women were monitored for weight gain, blood pressure, urine dipstick, glucose levels, other morbidities, compliance to antenatal visit, dietary intake, exposure to sunlight and presence of adverse effects. Twenty-four-hour dietary recalls were conducted to determine general eating patterns and assess dietary calcium and vitamin D intake. Compliance to vitamin D supplementation was assessed through self-reporting and evaluation of syrup bottles at each follow-up visit. After delivery, the newborn was assessed for neonatal abnormalities, gestational age and birth weight to identify preterm births and low birth weight.
Laboratory analyses
Maternal blood samples were collected at baseline and prior to delivery. At delivery, cord blood samples were obtained from the newborns. In the absence of cord blood samples, blood samples were collected from the newborns. The blood samples were used to assess serum calcium, phosphorus, alkaline phosphatase and vitamin D levels. The samples were transported to the Nutrition Research Laboratory at AKU for analysis.
Calcium, phosphorus and alkaline phosphatase were quantitatively determined colourimetrically using Roche Hitachi 902 chemistry analyser. Roche serum calcium (CA2), alkaline phosphatase (ALP2) and phosphorus (PHOS2) kits were used to estimate serum calcium, phosphorus and alkaline phosphatase levels. Two levels of quality control provided by Roche (Precicontrol levels 1 and 2) were used at minimum with each assay batch of calcium, phosphorus and alkaline phosphatase once every 24 hours. If a new cassette of reagent was used, a new calibration was performed using a calibrator for automated systems (CFAS) provided by Roche (Precicontrol ClinChem multilevels 1 and 2). A direct, competitive chemiluminescent immunoassay was used to measure serum vitamin D levels using LIAISON 25(OH)D Total Assay Kit on Diasorin LIAISON Immunoanalyzer, while quality control of the assay was monitored internally with every batch of the samples using 25-OH Vitamin D Total Control Set provided along with the assay kit. External quality control was assessed by participating in a Vitamin A Laboratory-External Quality Assurance offered by the Centers for Disease Control and Prevention (USA). The interassay coefficients of variations were 8.77% for serum 25(OH)D, 5.86% for serum calcium, 5.91% for phosphorus and 9.79% for alkaline phosphatase.
Data management and statistical analysis
Data were entered using databases and entry screens developed on Microsoft FoxPro. All data were double-entered for accuracy and quality control. Data were analysed using SPSS V.15. Descriptive statistics were reported through mean and SD for continuous variables and frequency tables for categorical variables and were adjusted for confounders. Means between groups were compared through an analysis of variance (ANOVA) test for continuous variables, and Pearson’s χ2 test was conducted to establish differences between treatment groups for categorical variables. A Fisher’s exact test was conducted for evaluating differences between groups with small sample size.26 An ANOVA test was also conducted to compare differences in mean serum vitamin D levels between baseline and endline between treatment groups. We conducted a multivariate linear regression to assess the impact of the intervention and confounding factors on serum 25(OH)D levels at the end of the study. Significance between groups was considered at p<0.05.
All participants were required to give informed written consent to participate in the study. Consent form was translated into local languages for better understanding. Other study ID numbers: PF8/0911.
Results
Baseline characteristics
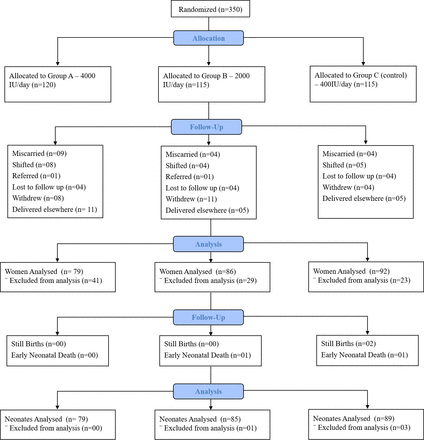
A total of 350 women were enrolled in the study and randomly assigned to a treatment group, with 120 women in group A (4000 IU/day), 115 women in group B (2000 IU/day) and 115 women in group C (400 IU/day) (figure 1). Two hundred and fifty-seven (73.4%) women completed the study. Socioeconomic characteristics were similar between treatment groups in terms of maternal age, maternal education and occupation status, husband’s education and occupation status, gestational age, and anthropometric measurements, and did not significantly differ across groups (table 1). Our data revealed that most households (93.1%) perceived that they received an adequate amount of sunlight, and that majority (96.3%) of women wore a veil or a burqa when outside their houses, likely due to religious and cultural norms. Moreover, the average time spent in the sun across all groups was 65.66±40 min/day, with no significant difference across groups.
Baseline socioeconomic and clinical characteristics of women participating in the study at enrolment, by study group
CONSORT diagram of participant enrolment, allocation, follow-up visits and analysis. CONSORT, Consolidated Standards of Reporting Trials.
Maternal and neonatal biochemical markers
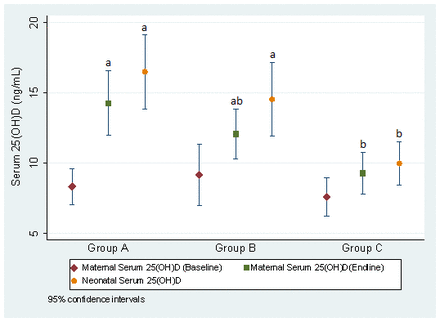
At baseline, mean biochemical markers did not differ across groups, except for phosphorus, which was higher in the control group (p=0.014). Maternal serum 25(OH)D concentrations were higher in group A compared with group C (14.0±9.6 ng/dL vs 9.8±7.2 ng/dL, p=0.002) at delivery, and neonatal 25(OH)D concentrations at birth were higher in groups A and B compared with group C (group A: 17.4±13.8 ng/dL and group B: 14.5±11.5 ng/dL vs group C: 10.2±7.1 ng/dL, p=0.006) (table 2). Group A saw a significant increase in both neonatal and final serum 25(OH)D levels compared with baseline, whereas only neonatal serum 25(OH)D was significantly higher than maternal baseline levels in group B (figure 2). Vitamin D deficiency (<20 ng/mL) was present in 336 (97.4%) women at baseline (group A: 116, 97.5%; group B: 109, 94.8%; group C: 107, 96.4%), with similar trends across groups (table 2). At endline, women in group A had the lowest vitamin D deficiency (75.9%) compared with those in group B (84.9%) and group C (90.2%; p=0.006). Unadjusted and adjusted serum 25(OH)D levels and prevalence of severe deficiency and deficiency in serum 25(OH)D at maternal baseline and endline are presented in online supplemental table 1. Vitamin D deficiency was significantly lower among newborns in group A (64.9%) and group B (73.7%) compared with group C (91.8%; p=0.001) (table 2). There were no significant differences reported between groups for other biochemical assessments. However, our study identified a marginally higher calcium deficiency among women across all groups right before delivery compared with baseline at the time of recruitment.
Supplemental material
Average and 95% CI of serum concentrations of vitamin D in women (baseline and endline) and their newborns across intervention groups. Different letters indicate significant differences between groups among the same population type. Maternal endline: groups A and C were statistically significant (p=0.003). Neonatal: groups A (p<0.001) and B (p=0.036) were statistically significant from group C.
Unadjusted maternal and neonatal biochemical markers* and maternal and neonatal biochemical deficiencies†
We evaluated the factors associated with serum 25(OH)D levels among participants at the end of the study using multivariate linear regression (table 3). Serum 25(OH)D levels were positively correlated with those in intervention group A (4000 IU/day) (β=4.16, p=0.002), with food group consumption (β=0.95, p=0.047) and with baseline levels of serum 25(OH)D (β=0.43, p<0.0001).
Factors effecting endline serum 25(OH)D (ng/mL) of women
Compliance to supplementation and participant dropouts
We further evaluated the overall compliance to vitamin D supplements among our study participants. The average compliance to supplementation across all groups was 79.4%±16.0, with no significant difference in compliance between study groups (table 4). A comparison between women who completed the study and those who dropped out is presented in online supplemental table 2. A total of 93 women dropped out of the study, with 41 in group A, 29 in group B and 23 in group C (figure 1). Details regarding study discontinuation are presented in figure 1.
Pregnancy and birth outcomes and exposure to vitamin D (compliance with supplementation and food frequency) among participating women
Pregnancy and birth outcomes
Pregnancy and birth outcomes did not significantly differ across the groups (table 3). There were 4 (1.6%) cases of pre-eclampsia and 11 (4.3%) cases of gestational diabetes reported. With regard to birth outcomes, there were a total of 74 (29.2%) preterm births in our study, 48 (19%) occurrences of low birthweight babies and 2 (0.7%) stillbirths, both in control group C. Moreover, there were two early neonatal deaths in our study, one each in groups B and C.
Safety outcomes
We did not encounter any episodes of hypercalcaemia or hypervitaminosis D in any of the participants in their prenatal or postnatal period or among the newborns. We also did not observe any serious adverse events or adverse events among participants across the study groups.
Discussion
Vitamin D deficiency has become a widespread global health issue, particularly among pregnant women in South Asian and Middle Eastern populations.27–29 To the best of our knowledge, this is the first individually randomised, controlled, dose comparison trial of vitamin D supplementation among pregnant women who experience vitamin D deficiency in Pakistan.
We found a vast majority of pregnant women (96.3%) were deficient in vitamin D at enrolment. The prevalence of vitamin D deficiency observed is alarming and the findings in our study are similar to other studies conducted in Karachi, Pakistan, which reported deficiency in over 90% of pregnant women.15 30 The recent NNS in Pakistan (2018) showed a deficiency of vitamin D among pregnant women of 79.7%, with a higher prevalence in urban areas.13 Another cross-sectional study reported that 88% of newborns were deficient in vitamin D, which aligns with our findings for newborns in the control group.31 Vitamin D deficiency has been highlighted an epidemic in South Asian populations, both in South Asian and Western countries.32 33 It has been speculated that this is due to the different genetic profiles of South Asians, but it has yet to be determined.34 Despite abundant sunlight in Karachi, low serum 25(OH)D could be attributed to the use of a veil/burqa among nearly all women in our study when outside their house, which has been associated with low absorption of sunlight and therefore low vitamin D levels in several studies conducted in Islamic countries.35–38 Moreover, the most abundant dietary source of vitamin D is oily fish, which is perceived expensive in Pakistan and not regularly consumed in the summer months. Another point to note is that, although many countries fortify milk and milk products with vitamin D, milk in Pakistan is mostly unpackaged and untreated with fortified minerals. Therefore, dietary practices in Pakistan do not provide sufficient vitamin D, which also results in calcium deficiency among women, as seen in our study. It has been speculated that vitamin D metabolism is altered in South Asians since they have higher 25(OH)D-24-hydroxylase activity, which increases degradation of 25(OH)D.39 However, further research is required to confirm these findings in populations residing in South Asia.
The recommended dose of vitamin D supplementation required during pregnancy remains debatable.40 We observed that supplementing women with 2000 IU/day was not enough to significantly improve their 25(OH)D concentrations at the end of the study. However, supplementing 4000 IU/day improved both maternal and neonatal serum 25(OH)D levels, but only reduced vitamin D deficiency in our population by approximately 20%. We also found that supplementing pregnant women with 4000 IU/day vitamin D3 was safe since no adverse events were identified among our participants and we did not observe hypercalcaemia or hypervitaminosis D. As per our knowledge, some studies have observed health outcomes with 4000 IU/day of supplementation and reported its safety and effectiveness.20–22 41–44 Our findings support previous studies mentioned above, which have shown that 4000 IU/day of vitamin D supplementation was effective in reducing vitamin D deficiency in pregnant women and newborns compared with supplementing 2000 IU/day or 400 IU/day.
We did not find clear evidence of the benefit of improved vitamin D status in pregnancy on health outcomes, possibly due to the small sample size and large number of dropouts. The relationship between vitamin D deficiency and GDM has been described frequently in the literature.45 Similar to a study conducted in neighbouring countries, our study also found an insignificant effect of vitamin D supplementation on GDM.46–48 The same effect of vitamin D was seen on the incidence of pre-eclampsia, where although groups receiving a higher dose of vitamin D presented fewer cases of pre-eclampsia, the overall occurrence of pre-eclampsia and the difference across the groups were not significant.47–50 Our study did not find any significant difference in the occurrence of preterm birth between intervention groups. Meta-analyses among systematic reviews have shown conflicting results for this outcome. A meta-analysis of three trials found a lower risk of preterm birth among women who received vitamin D supplementation, whereas another meta-analysis of 13 trials and 1 of 7 trials found no effect, which aligned with our study findings.12 51 52 For the occurrence of low birth weight, our results also show no difference in its prevalence among neonates whose mothers received vitamin D supplementation, which was seen in a meta-analysis of seven studies.51 Most meta-analyses conducted on randomised trials of vitamin D supplementation have shown an uncertain impact of supplementation on maternal and neonatal outcomes. Despite the debate over the impact of vitamin D supplementation, our study supports earlier findings, where administering a supplementation of 4000 IU/day did not result in congenital abnormalities, serious adverse events, or adverse maternal and neonatal outcomes.53 54
At the end of the study, approximately 75% of women who received the upper limit of 4000 IU/day still had vitamin D deficiency, which remains concerning. Correcting the insufficient status of all women of reproductive age, ideally prior to their pregnancy through loading doses followed by a maintenance dose, would be ideal in ensuring that pregnant women achieve vitamin D sufficiency.
Our study had limitations. First, our study findings may not be generalisable to a global population due to the severity of vitamin D deficiency in Pakistan compared with the rest of the developed world.44 The study was single-centred and was conducted in urban areas of Karachi, Pakistan, which is not reflective of rural populations and other provinces. Second, after supplementing with 4000 IU/day approximately 75% remained deficient at delivery and our results also did not produce significant differences in pregnancy and birth outcomes across different study groups. This is possibly due to our underpowered sample size towards the end of the study. Although we recruited more participants than needed, about a quarter of them did not complete the study. We had a high dropout rate, with almost 41 from group A, which has overshadowed the impact. Checking levels of vitamin D at 2 months after initiation of the study would have been a better option, as reported from other studies, rather than at the time of birth. Third, we could not use any validated form to measure compliance, nor was it directly observed; it was assessed through self-reporting and evaluation of empty syrup bottles. Non-compliance to regimens could be a reason for higher rates of deficiency at endline. Fourth, there might be low bioavailability of vitamin D in the syrup used in the present study, which could be due to low fat content or biochemical formation. Although the drug was stored at room temperature as advised, issues of bioavailability cannot be ignored. Similarly, absorption of vitamin D is low in certain gastrointestinal disorders and malabsorption syndromes, which was not evaluated. There are not many studies on pregnant women from South Asian countries and the role of genetic make-up is still not understood, which could be an important factor for low uptake and needs to be explored in further studies. Another limitation of our study was that we did not assess calcium or vitamin D intake through diet and used a 24-hour recall to assess if there was a difference between groups in dietary consumption.
Conclusion
The evidence provided in our study indicates that vitamin D supplementation of 4000 IU/day was more effective in reducing vitamin D deficiency among pregnant women and improving serum 25(OH)D levels in mothers and their neonates compared with 2000 IU/day and 400 IU/day. Moreover, all formulations of supplementation are safe as no adverse events were reported in our study. Further studies would benefit by following up with newborns of mothers enrolled in supplementation trials to identify long-term outcomes and benefits of supplementation.
Data availability statement
Data are available in a public, open access repository. The datasets used for the article and the study is available from the corresponding author on request.
Ethics statements
Patient consent for publication
Ethics approval
Ethical approval for the study was obtained from the Aga Khan University Ethical Review Committee.
Acknowledgments
We would like to thank the trial participants for their voluntary participation in the trial. We are also thankful to data collectors, nurses and the trial staff who performed routine activities to complete the trial and especially Ms Farahan Tabassum for leading the field team.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Twitter @sajidsoofi
Contributors SN was the principal investigator of the study. SN and SBS conceptualised the study idea and design and interpreted the data. SN and MB wrote the first draft of the manuscript. AR and FS contributed to data management and analysis. SSR oversaw the preparation of vitamin D formulations at the pharmacy. KB was responsible for biochemical assessment at the laboratory. JI, LS, AH and SBS critically reviewed the manuscript. All authors contributed to manuscript review and approved the final manuscript.
Funding This study was funded by the University Research Council at the Aga Khan University, Pakistan.
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.